PFAS: Working toward a sustainable future while protecting patient care
As an industry, we recognise the concerns over the environmental impact of Per- and polyfluoroalkyl substances (PFAS). We support the ambition of achieving a healthier and more sustainable environment as part of our wider commitment to invent, produce and distribute new medicines and vaccines in a safe and environmentally responsible manner. Companies are already taking significant actions to find safe and sustainable solutions for the future.
PFAS are an integral part of medicines development. They are used throughout the entire lifecycle of a medicine and vaccine, from the early stages of research and manufacturing all the way to the final packaging and delivery devices. These materials and uses have been tested and approved under pharmaceutical regulations.
PFAS are a broad and diverse group of fluorinated materials, environmental and health concerns associated with certain PFAS have led to a proposal for a European-wide ban on PFAS; however, given that REACH restrictions apply to approved medicinal products, the proposal has significant implications for pharmaceutical products, packaging and manufacturing operations.
Replacing all materials that contain PFAS in a highly regulated sector like pharmaceuticals is challenging and requires an appropriate transition period to be set by the European Commission to allow adequate time to seek alternatives and adapt processes to minimize identification of suitable alternatives and an impact on patients.
A sector-wide industry survey illustrates the potential scale of the impact of an EU universal PFAS restriction on medicines:
- Affected Medicines Companies identified 1,922 active substances and at least 47,677 global marketing authorisations that could be in scope.
- Essential Treatment Over 650 medicines on the WHO Essential Medicines List would be affected and potentially not accessible anymore, many of which overlap with national critical medicines lists in the EU. These are used to treat serious and long-term conditions like cancer, diabetes, HIV/AIDS, depression, malaria, multiple sclerosis and inflammatory diseases.
Policymakers must balance environmental concerns with the need to safeguard patient access to medicines, protect Europe's innovation capacity and preserve strategic autonomy in healthcare.
The Reality of Replacement
Industry data shows that more than 90% of active substances rely on EU manufacturing operations that depend on these materials, often with no suitable alternatives currently available.
Substituting just one active substance containing a PFAS goes far beyond reformulation. It would require discovering and developing a completely new medicine—a process that typically takes 12 to 22 years and demands an investment of at least €2.3 billion.
Pharmaceutical manufacturing also relies on PFAS. For many applications, no alternatives exist that perform equally well, ensuring sterility, purity and stable processes. If a broad ban forces production to shift outside the EU, Europe's strategic autonomy in healthcare would be severely weakened without actually reducing global PFAS use.
The economic fallout of a broad ban is also significant, with estimates pointing to a potential loss of €31 billion in annual revenue in the EEA and an estimated 22,500 job losses2. Europe is a net exporter of medicines. Without the pharmaceutical industry the EU trade balance would go from a surplus of €133 billion to an €88 billion deficit.3
PFAS are an integral part of medicines development
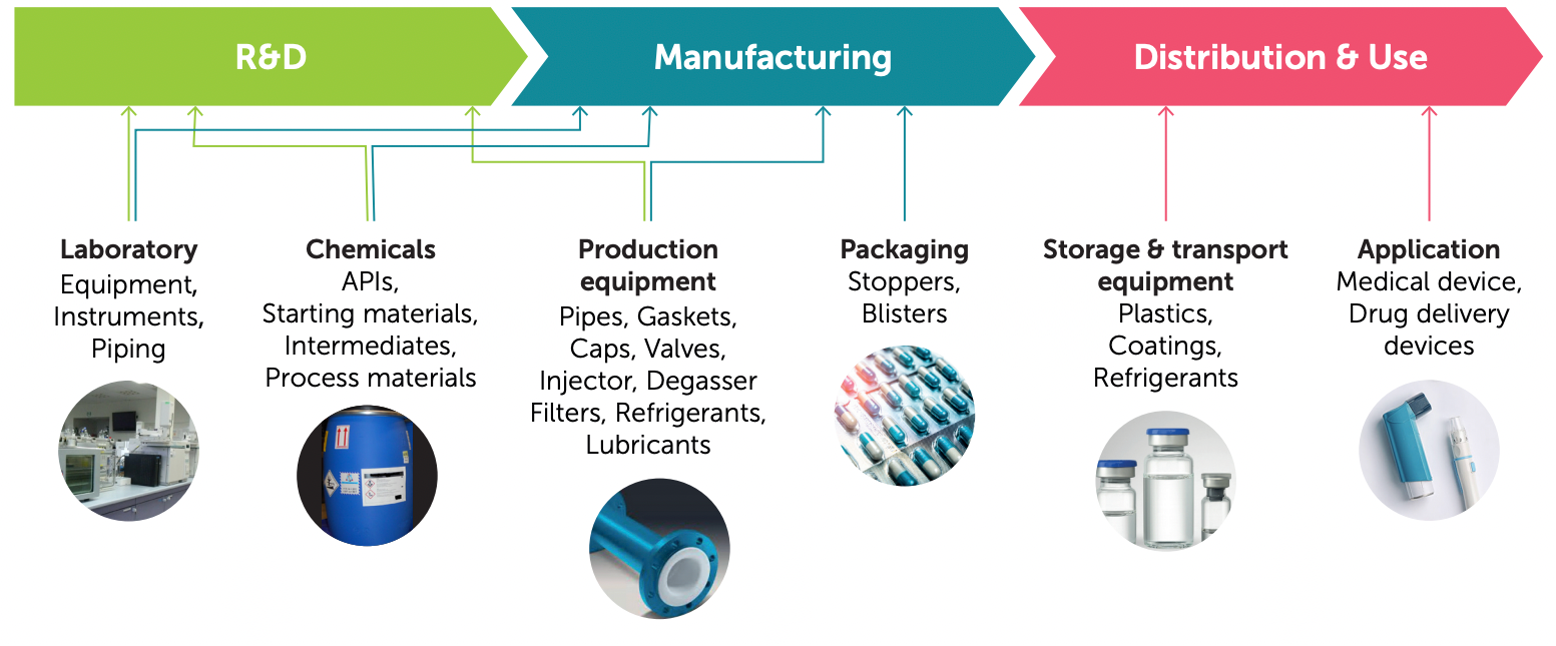
Why PFAS matter in a medicine
Hundreds of approved medicines contain fluorine. Fluorine makes medicines work better. It acts like a precision tool, helping drugs to be:
- More stable: They don't break down easily.
- Longer-lasting: They stay in the body longer, often requiring less frequent dosing.
- More effective: They are better at reaching their target in the body, often with fewer side effects.
As a result, the medicine has a longer-lasting effect at lower doses, which minimises emissions. Just like the health effect, the environmental impact of medicine has to be assessed individually, which in pharmaceutical legislation is conducted through Environmental Risk Assessments (ERA).
Why PFAS matter in R&D
PFAS are present across the lifecycle of a medicine, including in research and development. The survey identified 169 APIs undergoing process development in EU manufacturing facilities. Without appropriate derogations for product and process-oriented research and development, clinical trials are impacted.
High-Value Investment: the research-based pharmaceutical industry invested €55 billion in research and development in 2024. It directly employs around 130,000 people in research roles out of a total workforce of nearly 950,000.
Why PFAS matter in medicine manufacturing4
Fluorinated chemicals play an important role in the manufacture of many medicines. For example, trifluoroacetic acid (TFA) is necessary for producing peptide therapeutics used to treat diseases such as diabetes, HIV, growth disorders and osteoporosis. Currently, no viable alternative exists for this process, and replacing TFA would require extensive regulatory approvals from health authorities worldwide.
Fluoropolymers are also widely used in pharmaceutical manufacturing equipment (reactors, pipes, seals and filtration systems) because they are strong, stable and meet strict quality and safety standards.
Supply Chain Disruption: Replacing these materials is complex. Any modification to approved manufacturing processes requires testing and regulatory review to ensure medicines remain safe and effective. A restriction could halt production once existing stocks are depleted, potentially disrupting the supply and access to medicines across Europe and globally.
Emission of persistent chemicals are a serious environmental concern. However, manufacturing materials do not become part of the product, and the manufacturers conform to air and wastewater limits as well as waste regulations. Emission limits can be adapted to address any concerns.
Why PFAS matter in packaging & delivery devices
Patient safety: PFAS are used in specific packaging components and drug delivery systems (like inhalers), where they provide essential barrier and performance properties.
An industry survey identified 276 EU-authorised products using polymeric PFAS in packaging and 98 EU products incorporating PFAS-containing delivery devices.
Lengthy process: Replacing these components is complex. For example, reformulating a single inhaler product with an alternative propellant typically requires six to ten years of development. These timelines reflect patient safety requirements. They cannot be compressed without risk.
Packaging specific initiatives and regulations address environmental concerns and balance different aspects such as minimising waste and single use plastics, protection of the product, recyclability, and emission of contained substances including PFAS.
Why PFAS matter in quality control and pharmacopoeial standards5
Quality assurance systems rely on PFAS-based materials and reagents. More than 170 European pharmacopoeia monographs reference PFAS reagents, and over 90% of EDQM reference standards, more than 2,500 in total, are supplied in containers with PTFE-coated closures. These methods are legally mandated and internationally harmonised. Modifying them requires formal pharmacopeial revision processes and coordinated regulatory action, which takes many years.
Cold chain maintenance (F-gas): The maintenance of low temperature and humidity control of processing and storage areas (air processing units) are essential in vaccine production because it preserves the stability, potency, and safety of temperature-sensitive biological components.
Environmental responsibility and emissions management
Companies are actively working to reduce their environmental footprint by exploring safe and sustainable alternatives where feasible and collaborating with suppliers to minimise PFAS use across manufacturing and supply chains. In parallel, companies are actively contributing to major European public–private research initiatives aimed at better understanding and reducing the environmental impact of pharmaceuticals, including PFAS.
Active substances are already subject to environmental risk assessment as part of a strengthened marketing authorisation process. For many industrial uses of fluoropolymers, emissions can be managed through responsible waste handling and end-of-life treatment.
Through projects such as IMI PREMIER6, IHI PHARMECO7, and IHI ENKORE8, industry partners are collaborating with academic institutions, regulators, and technology developers to advance environmental monitoring, improve understanding of pharmaceutical emissions, and develop innovative solutions for more sustainable manufacturing and waste management.
IHI PFAS project (launching 2026) is a €44.4 million Innovative Health Initiative project bringing together 97 organisations and more than 250 experts across 13 EU countries. Its goal is to better understand PFAS use in healthcare, develop alternatives, improve monitoring and support regulatory alignment.
New Monitoring and Treatment Methodologies: Many sites are strengthening environmental monitoring and upgrading wastewater treatment to track and remove PFAS, often achieving levels already below national limits.
Responsible Management and Emission Control: Emissions can be effectively managed through responsible waste handling and end-of-life treatment, such as mandatory waste incineration requirements under the Good Manufacturing Practice (GMP). Industries are reinforcing and improving technologies to ensure essential uses have minimal impact on the environment.
The industry proposes that targeted, emission-focused measures are more proportionate and effective at solving the environmental challenge than blanket bans on essential uses. Science-based collaboration should drive a future innovative EU landscape that ensures a smooth and sustainable transition, and align as far as possible with global initiatives to create a level playing field and minimize overall emissions.
References
- 1 https://www.efpia.eu/media/52ipvgfi/annex-1-efpia_sea_pfas_final.pdf
- 2 https://www.efpia.eu/media/52ipvgfi/annex-1-efpia_sea_pfas_final.pdf
- 3 EFPIA Statement Feb 2026 – European Pharma key to strengthening EU Competitiveness
- 4 https://www.efpia.eu/media/jc1lcupo/annex-3_industrial-use-of-fluoropolymers-in-pharma-manufacturing_final.pdf
- 5 EDQM presentation, Sustainability session, DIA Europe 2024 + EDQM submission to the public consultation at ECHA on the universal ban on PFAS proposal, September 2023
- 6 https://imi-premier.eu/
- 7 https://pharmeco.eu/
- 8 https://enkoreecohealthcare.eu/