10 things I’ve learned about IP policy
26.04.22
At the end of last year, I was lucky enough to land my first real job at EFPIA. I say lucky, because this is not an ordinary job – straight in at the deep end to staff the IP working group. Even though I have a background in European law, this was overwhelming at first, but at the same time, gave me an amazing opportunity to learn from leading international IP experts.
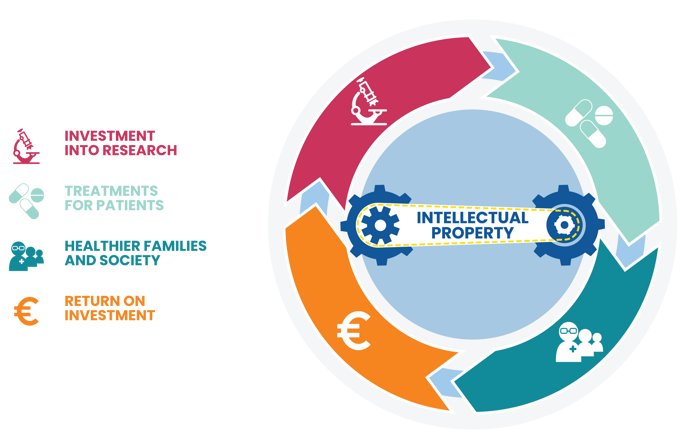
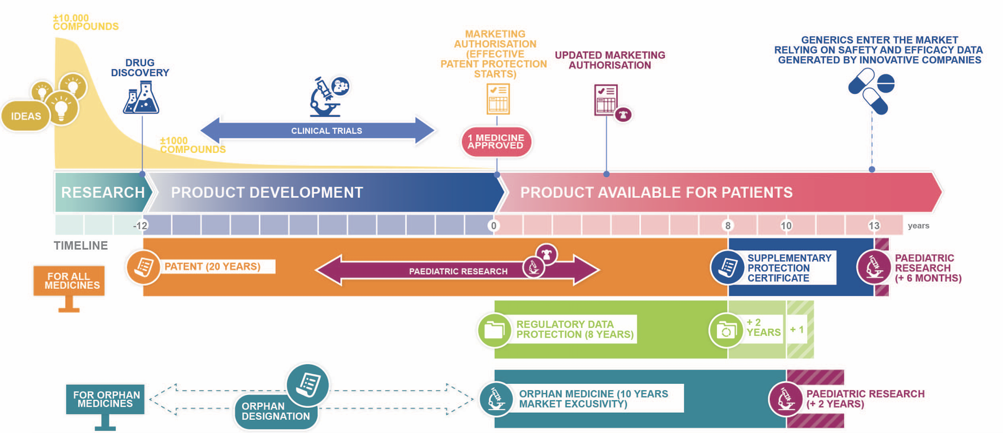
They sparked my new passion for IP law. I quickly came to view IP as a valuable asset for society, creating possibilities to share, license and incentivize inventions.
Here are the top 10 things I learned about IP law since I started working at EFPIA: