Why aren’t we there yet? The role of partnerships in unleashing patient-centred health data (Guest blog)
In this blog, Meni Styliadou from Takeda and Francesco Patalano from Novartis, both EFPIA members, explains how the H2O public-private partnership will enable the collection of patient-centred data, contributing to making this crucial component of value-based health systems a reality.
Today, the health outcomes of this enormous patient population are not captured at scale and therefore largely unknown, which should be a concern for everyone. [see figure 1] First and foremost for patients, but also for all stakeholders and policy-makers that want to make health systems more patient-centred. In many cases, these PROs are being gathered in some form, because it is commonly understood that they inform high-quality patient care, including through enhancing the dialogue between the patient and the treating physician.

But they are being gathered on a small scale, usually kept within institutions and almost never shared between health care providers or across borders. Furthermore, they are not utilised for decisions and policy-making at regional and national level of health systems., As a result, this limits their usability and potential valuable insights that a greater scale might allow.
What truly matters for patients? The answer to this simple question varies from person to person. Therefore, integrating patient related outcomes in any healthcare decision-making process is crucial. We believe that gathering patient related outcomes and harnessing the patients’ lived experiences represent the building blocks of future healthcare. They enable informed decisions and more efficient advocacy efforts that, at the end of the day, lead up to our ultimate common goal: high-performant patient care. Valentina Strammiello, Director of Programmes, European Patients’ Forum
It’s clear that so many parts of the health care system are calling out for this data. In addition to improving the care of the individual patient, it could also be utilised for assessing and comparing treatment options and care pathways, provide evidence for outcomes-based payment models and inform overall healthcare policies and performance assessment to name a few examples. If this data could be captured and utilised at a larger scale than today, there is huge potential for improving health system efficiency, improve patient and population health outcomes, boost research, inform Health System Performance Assessment and monitor the patient-centredness of health systems. [See Figure 2.]
As an industry, we have made many forays into exploring this source of important data, but no-one has found a large-scale, sustainable solution to gathering PROs beyond the limits of clinical trials. There is a reason for this.
Figure 2

The primary aim of healthcare should be improving the outcomes of patients. The focus in outcome measurement has recently shifted from clinical variables only to the integration of clinical and patient-reported information. Considering outcomes important to patients can change healthcare substantially. Patient-centred public-private partnerships like H2O can accomplish such systemic changes. Prof. Dr. Tanja Stamm, Head of Section for Outcomes Research, Deputy Head of Center for Medical Statistics, Informatics and Intelligent Systems at the Medical University of Vienna (H2O Scientific Co-Lead)
It’s because the pharmaceutical industry cannot find the solution alone, no more than the regulators or the hospitals or technology companies could. The data is ultimately reported by and belongs to patients, so for any project to be sustainable and successful it must not only include but be actively driven by patients and patient representatives.
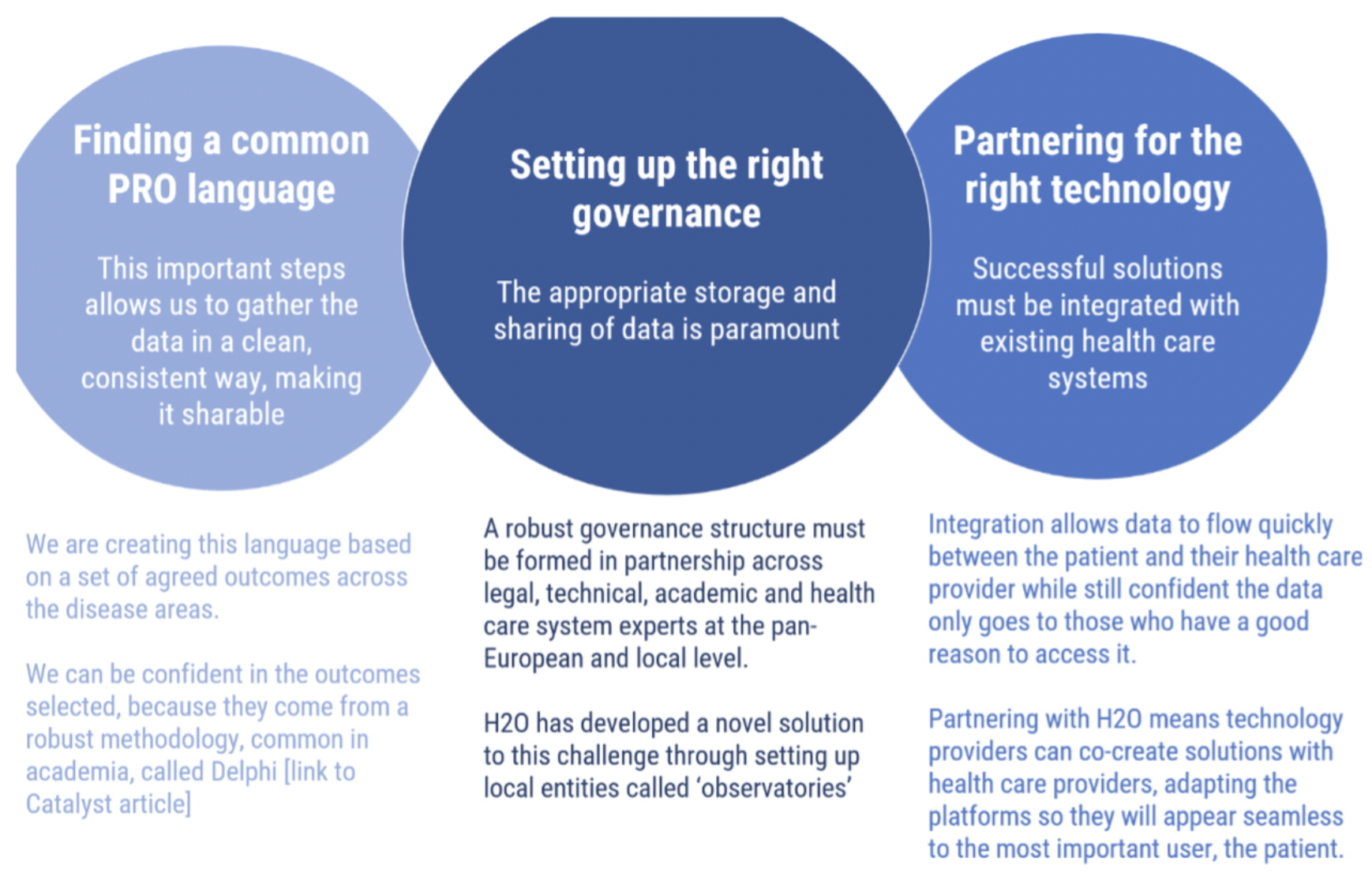
Sharing PRO data on this scale calls for a unique alignment of multiple conditions. Technical platforms and guidelines aren’t enough. For patients and their health care providers to feel it is both useful and safe to share this deeply personal information, trust must be built at every level.
It must be clear that the governance, the technology and the methodology fully respect ethical principles, are expertly designed and lead to widely accepted standardisation. Many organisations are doing inspiring and excellent work in bringing PROs into the foreground, but most do not have the capacity to draw on the wide partnership needed to take on the challenge of standardisation of PROs and sharing data on a large scale.
The Health Outcome Observatories project (H2O) is the first large-scale attempt to build the necessary trust needed to unleash the potential of PROs. [See figure 3]
This addition to our partnership takes us even deeper into the health care system, not only to explore how PROs might be built into value assessments of healthcare interventions but also to find out what the regulators want to get out of the data. It’s these kinds of insights which will help H2O build the most effective approach to gathering and sharing PRO data, and this is only made possible through trusted partnerships.
H2O is an IMI-funded initiative, subject to all the scrutiny and due diligence required of a publicly-funded initiative [see figure 4]. This also means that the H2O work belongs to the public. Within the pharma industry, our experience with COVID-19 has demonstrated that we must open our doors to allow innovation to flourish.
Multiple EFPIA members are involved in what is shaping up to be one of the biggest leaps forward in incorporating the patient voice in the care pathway.

Please follow us on LinkedIn for the latest news. If you would like to find out more, contact us via our author links.
About the authors
- Meni Styliadou is a Distinguished Fellow in the Takeda Data Science Institute and the Project Leader of the H2O project which has received funding from the Innovative Medicines Initiative 2 Joint Undertaking under grant agreement Nº 945345-2, and also receives support from the European Union’s Horizon 2020 research and innovation programme and EFPIA and Trial Nation and JDRF.
- Francesco Patalano is Head of Cross-Functional PROs and Pediatric Centers-of-Excellence in Novartis Pharma AG and an Executive Committee member of the H2O project.
DISCLAIMER: this article reflects only the authors’ views. Neither IMI, nor EFPIA, nor the European Commission, nor the JU are liable for any use that may be made of the information it contains.
https://www.ihi.europa.eu/