Turning EU excellence in science into commercial success could add more than €120billion to EU economy in 10 years
New research provides the first comprehensive overview of the EU’s attractiveness as a destination for pharmaceutical investment.
New research for EFPIA by Charles River Associates provides the first comprehensive overview of the EU’s attractiveness as a destination for pharmaceutical investment, benchmarked against its global competitors: US, China, UK and Switzerland.
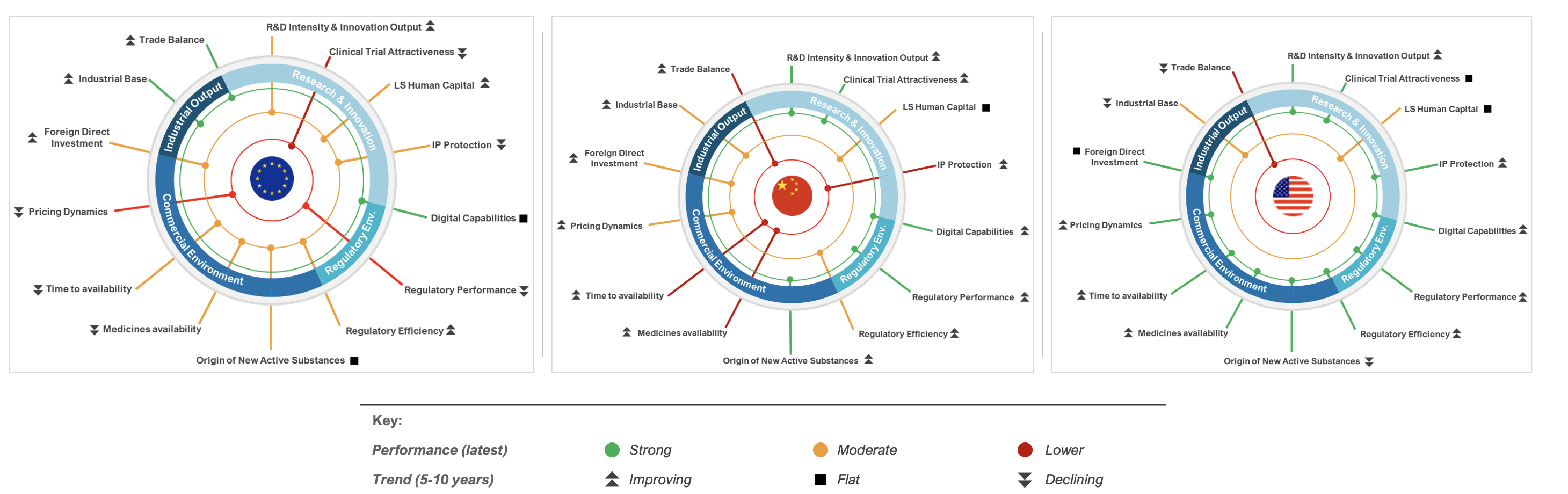
Published today by EFPIA, Assessing Europe’s Competitiveness as a Location for the Life Sciences Industry analyses 20 performance indicators across four key areas critical to attracting investment: research and innovation, regulatory framework, the commercial environment and industrial output – Europe’s complete life-sciences ecosystem.
The data underlines Europe’s continued leadership in early-stage scientific excellence, advanced digital capabilities, and pharmaceutical manufacturing, supported by a strong industrial base and trade performance.
However, the analysis also shows that Europe struggles to translate scientific excellence into research investment, clinical trials and new medicines. All of which deliver better health and economic growth.
As policymakers seek to strengthen Europe’s competitiveness, this new research helps inform the policy decisions that will shape the future of an industry integral to Europe’s health, economy and security. Each year pharmaceutical companies invest €55 billion in EU research and development, support around 2.3 million jobs and generate more than €366 billion in exports. Without the pharmaceutical industry, the EU’s overall trade balance would shift from a €133 billion surplus to an €88 billion deficit.
At the same time, recognising the strategic importance of life sciences, global competitors are moving further and faster to attract pharmaceutical investment. If Europe can match, or even exceed the pace and scale of this ambition, the report quantifies what the EU has to gain by closing the gap on its nearest rival:
- Closing the gap in industry R&D investment alone could generate an additional €105 billion in investment over the next decade.
- Increasing Europe’s share of global clinical trials could add nearly €18 billion to the European economy, create around 82,000 jobs and enable 158,000 more patients to participate in trials.
- Strengthening regulatory pathways and the broader innovation ecosystem could also accelerate the development of new medicines, with the potential to bring more than 200 additional new active substances (NAS) to patients and increase the number of medicines originating from Europe.
Fulfilling this potential requires bold and decisive action to build on Europe’s undoubted strengths and address the issues identified in the report that are standing in the way of Europe realising its ambition to be a world leader in life sciences.
Commenting on the data, Stefan Oelrich, EFPIA President and President of Pharmaceuticals at Bayer AG said: “Europe has the science, the skills and a strong industrial base, but we’re being outpaced by China and the United States on investment, clinical trials and advancing novel medicines. Europe’s challenge is to close the gaps identified in the report to create an environment that attracts investment, accelerates translation into new treatments and ensures day-one access for Europeans. If we’re willing to act to close the gap, we don’t just boost growth; we secure Europe’s health resilience. The choice is ours.”
Nathalie Moll, EFPIA Director General said: “This report shows both the scale of the opportunity for Europe and the areas where urgent action is needed to close the competitiveness gap on other regions of the world.
With the right decisions, Europe can build on its strong scientific foundations and industrial capabilities to secure a more competitive and resilient life sciences ecosystem. Strengthening Europe’s attractiveness for pharmaceutical investment will not only support economic growth and security but also ensure that European patients benefit more quickly from the next generation of medical breakthroughs.”
Key findings from the report:
Taken from the report, below is a brief summary of Europe’s strengths and weaknesses across a number of the indicators.
EU Strengths:
- The EU shows strong manufacturing investment growth (15% CAGR 2018–2022, surpassing China's 11%) and a persistent trade surplus – the largest contributor in Europe – reflecting resilient supply chains and export capabilities.
- The EU performs strongly in the percentage of medical sciences publications which are amongst the most highly cited (top 1%) globally.
EU Moderate performance:
- The discovery of new active substances (NAS) for drugs has fallen by 20% in the EU; in China it has leapt 470%.
- EU intellectual property protection exceeds only China.
- Origin of new medicines. China now leads the EU and the US as the originator of New Active Substances (NAS) - from 4 NAS in 2018 to 28 in 2024. NAS from China are increasingly being launched in the US and Europe.
- The launch of new medicines: On average, EU countries achieved a launch rate of 39%, substantially lower than the US with 85%. Disparities persist among leading EU performers, for example in Germany (61% launched overall and 44% within a year); compared to France, (52% launched overall and 23% within a year). Germany tops Europe but remains well below the US.
EU Weakness:
- EU Patent applications grew modestly (6% 2014–2024); China's 170% surge highlights the EU's relative stagnation. Trends indicate that the EU is not capturing a proportionate share of mobile innovation capital relative to global peers.
- Regulatory approval timelines have improved (430 days in 2024, down from 464 in 2015) but remain longer than in China (390 days) and the US (356 days).
- The EU’s performance in spending on pharmaceuticals is 1% of its GDP, compared to China (1.8%) and the US (2.0%)
- More than 20 EU Member States apply cost-containment tools such as mandatory rebates, mandatory sales taxes and claw-backs, while the US and China rely on more limited rebates or discounts. These mechanisms are widely reported to deter pharmaceutical investment.
- Every country in the EU, apart from Spain, is losing clinical trial share. China’s modernising society and population size make its growth ‘unsurprising’, says the report. The EU’s loss of market share is most concerning, with global share down from 22% in 2013 to 12% in 2023.
